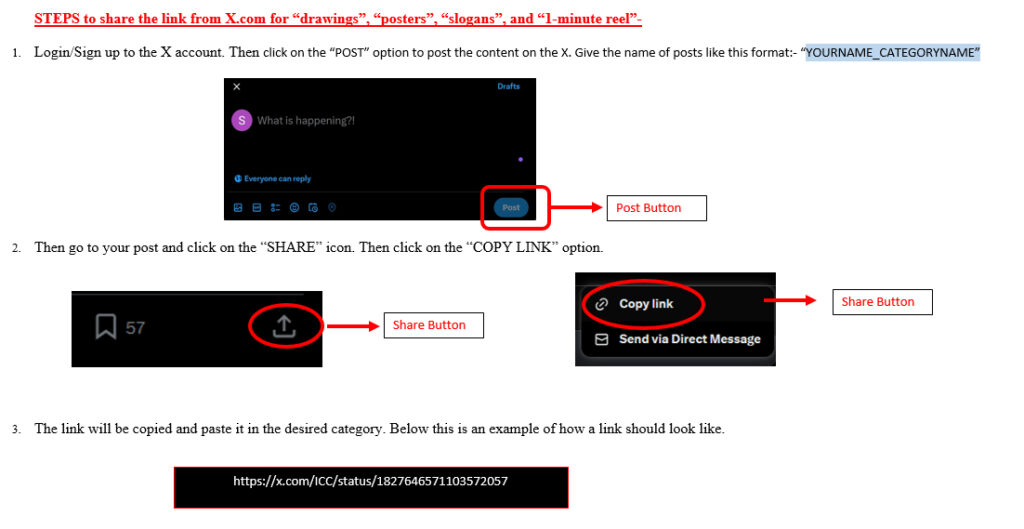
Academics
| Degree | University/Institution |
|---|---|
| P.hD. | University of Hyderabad, Hyderabad, India |
Work Experience
| Position | University/Organisation | Period |
|---|---|---|
| Scientist E | Institute of Life Sciences, Bhubaneswar | Nov 2020 onwards |
| Scientist D | Institute of Life Sciences, Bhubaneswar | Nov 2015 – Dec 2019 |
| Project Scientist | University of California, San Diego, USA | 2011 to 2015 |
| Post-Doctoral Fellow | University of California, San Diego, USA | 2008 to 2011 |
Awards & Recognition
| Details |
|---|
|
Research
| Details |
|---|
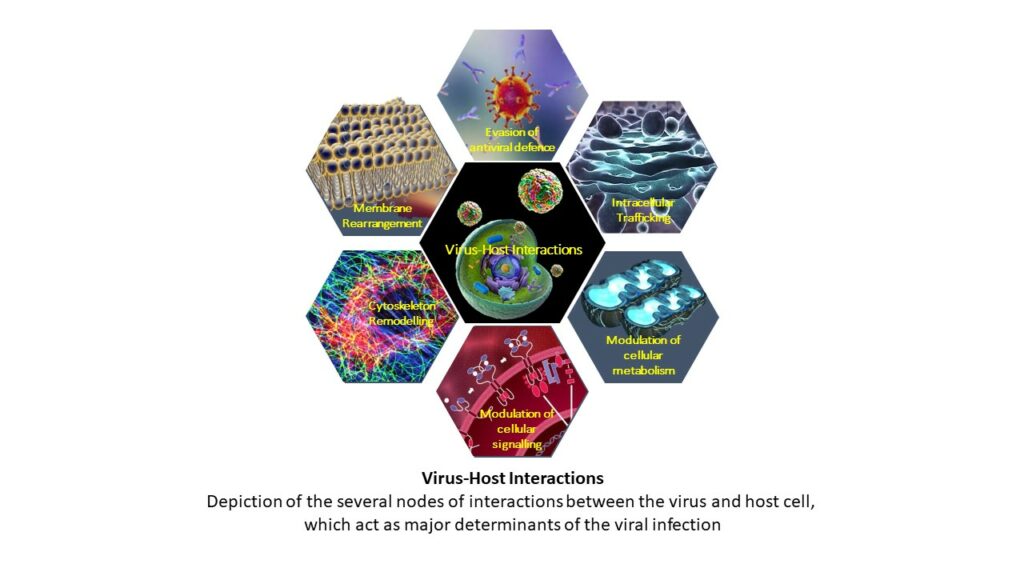
Viruses are obligate intracellular parasites with limited genome and thereby rely on the host cell machinery for their proliferation. Thereby viruses have evolved molecular strategies to escape the cellular innate immune surveillance and to usurp/hijack the host cell signaling to create a microenvironment conducive for viral dissemination. Most often the virus-mediated alterations to the host signaling or the response to infection paves way for the pathogenesis of the viral disease. Understanding these interactions will help in identifying the molecular mechanisms that drive viral diseases
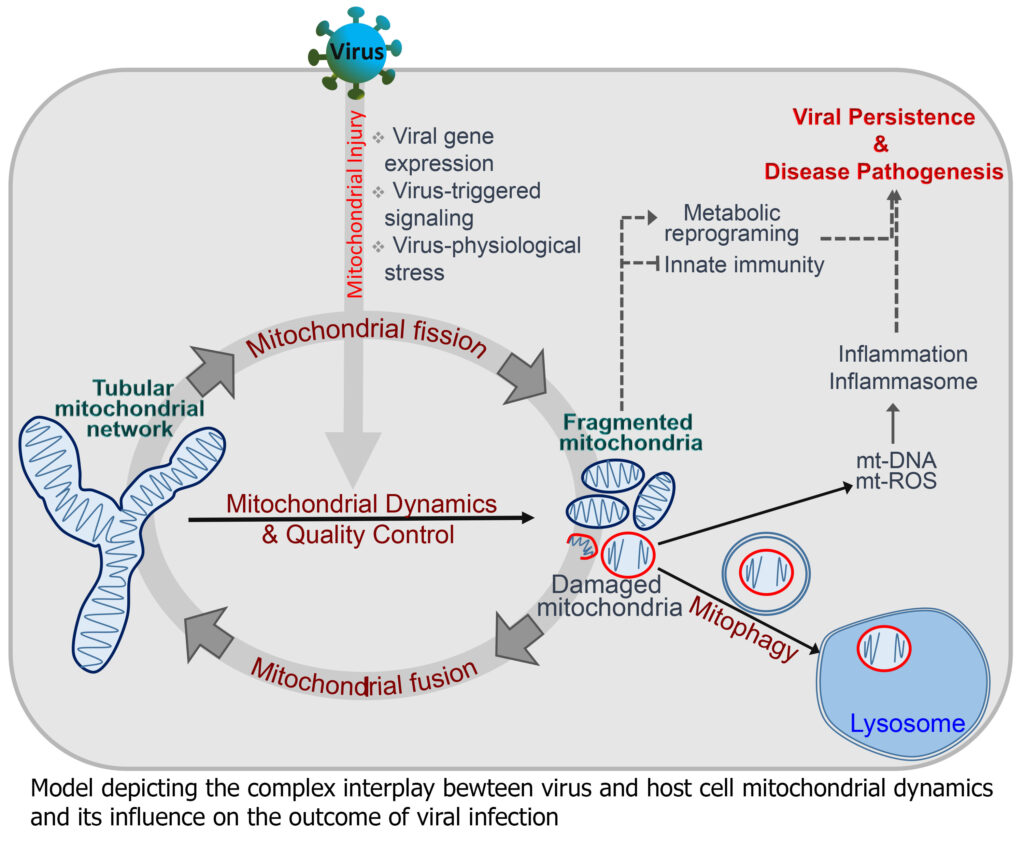
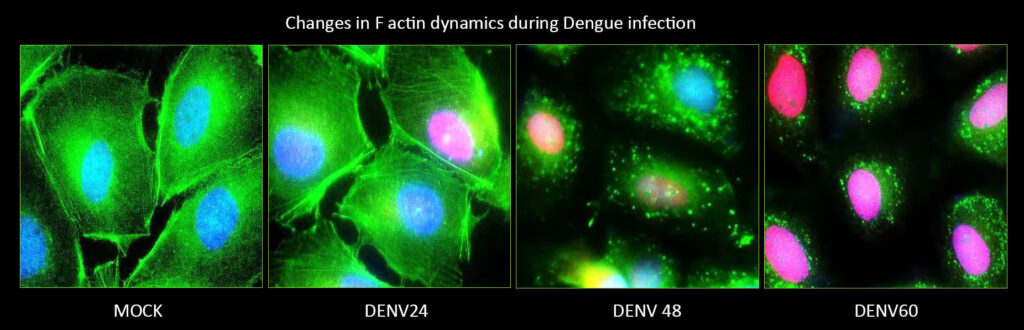
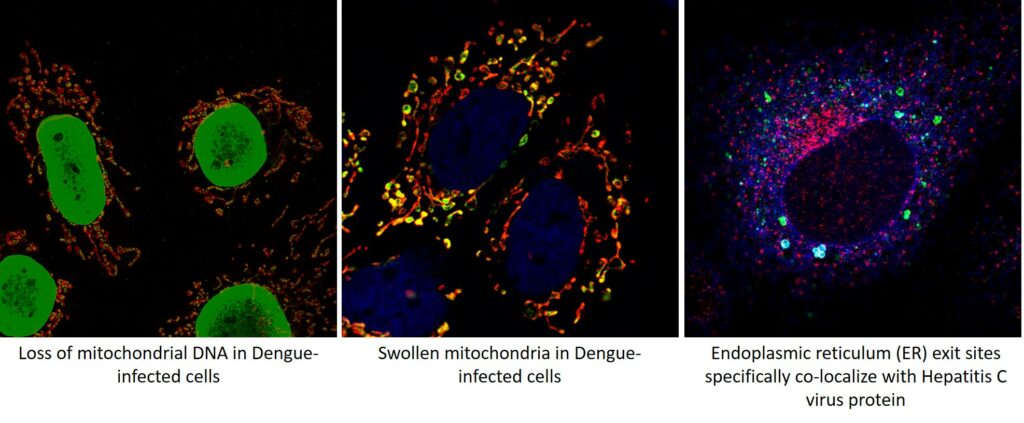
The cellular mitochondria, membranes and cytoskeleton are primary targets of many viruses. Mitochondria serve as the hub for many cellular events and mitochondrial dynamics is considered an integral cellular process with implications in cellular homeostasis, metabolism, inflammation and innate immunity. Thus many viruses target/exploit the host mitochondria to escape cellular defense mechanisms and promote viral proliferation. The physiological perturbation associated with viral infection also affect the host mitochondria.
The complex interplay between the virus and the cellular mitochondria and the subsequent consequence on mitochondrial- metabolism, antiviral and inflammatory signaling governs the outcome of viral infection and viral disease pathogenesis. The complex interplay between the viruses and the host machinery or organelle such as mitochondria is an ever expanding and unexplored area. Characterizing these interactions will unravel the molecular cues that promote viral dissemination and disease pathogenesis. Currently the lab is working with the flaviviruses, Dengue virus (DENV), Hepatitis C virus (HCV) and Japanese Encephalitis virus (JEV). During the pandemic we have also worked towards COVID-19 research and isolated local circulating strains of SARS-CoV2 and are conducting studies for repurposing of approved drugs against SARS-CoV2. We are also in the pursuit of screening compounds effective against DENV and JEV. In future we will also attempt to characterize the virus-host interactions in the pursuit of identifying therapeutic targets with pan-viral potential.
The major focus areas of the lab are;
|
Publications
| Details |
|---|
2022
2021
2020
2018
2017
2016
2015
2014
2013
2012
2011
2010
2007
Book Chapters
|
Group
| Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
PhD students:
Postdoctoral Fellows:
Project Fellow: Sabyasachi Pattnayak Lab Technician: Biswajita Prusty Past Members: Ruthu Nagraj (JRF), Subham K Sahoo (Lab Technician), Debjani Tarapdhar (N-PDF), Leela Kirshna Bankapalli (PDF), Subashish Samantray (Project Fellow), Poornima Kokavalla (SRF), Preethy V Kumar (JRF)
|
Grants
| Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Contacts
| Address | Fax | Office | |
|---|---|---|---|
| gulamsyed@ils.res.in | Institute of Life Science, NALCO Square, Chandrasekharpur, Bhubaneswar 751023, Odhisa | 0091 674 2300728 | 91-674- 2304287 (office), 91-674-2304318 (Lab) |
Highlights
| Details |
|---|
|
Positions
| Details |
|---|
Students who are interested in pursuing research & training in the field of virus-host interactions and molecular virology are welcome to pursue suitable programs in the Virus-Host Interaction Lab
|